  
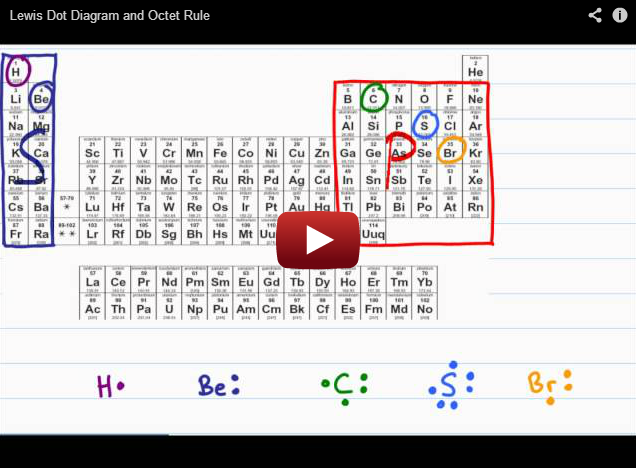
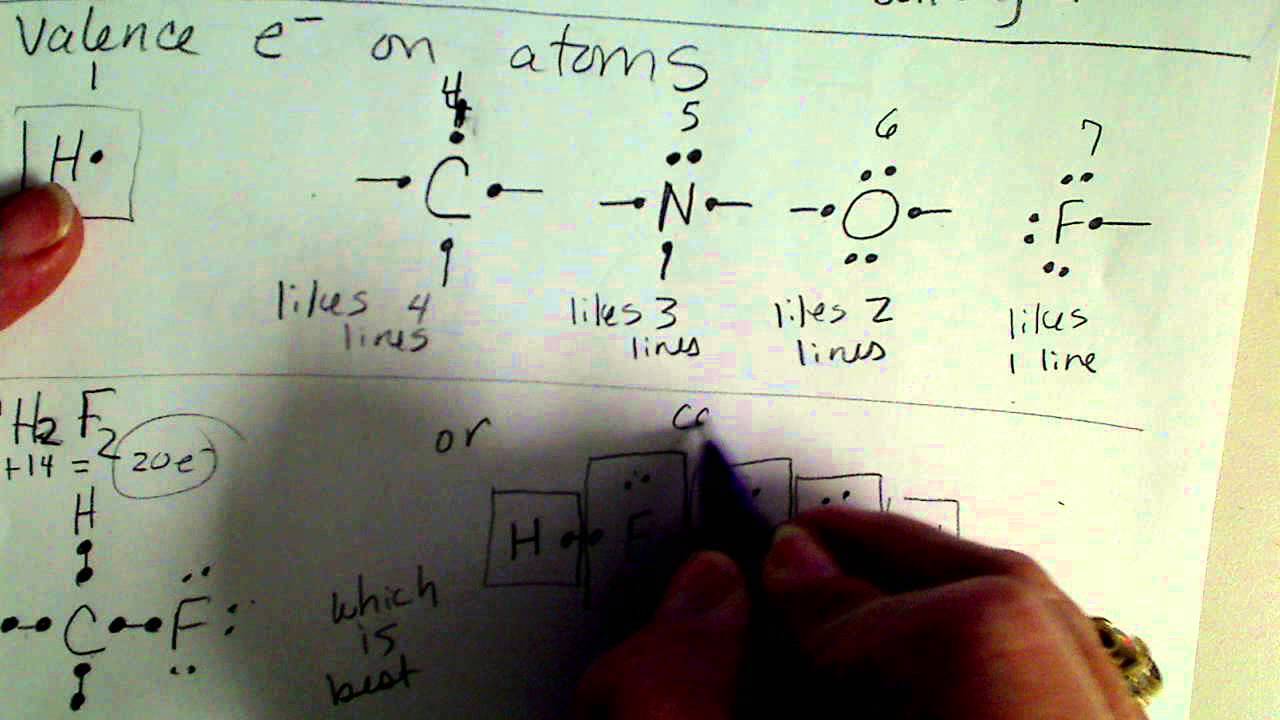
Since the molecular formula is O 3, we know there are 18 valence electrons (oxygen has six valence electrons as 6 x 3= 18). To find the resonance structure of ozone, we will draw the lewis structure of ozone. In chemistry, it is one of the most powerful oxidizing agents as it has a large standard reduction potential. The reason it is named “the ozone layer” is because of its high concentration of ozone, which absorbs the majority of the sun’s ultraviolet radiation. You have probably heard of the ozone layer in the earth’s stratosphere.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
